The Human Cytomegalovirus (HCMV) is the leading cause of viral birth defects. HCMV Pentamer Complex is considered a key target candidate for vaccine therapies. How HCMV avoids humoral immunity is not clear. New research into the elusive virus reveals the surprising role of the host cell’s endoplasmic reticulum to aid viral infection.
Protein Description 5VOB
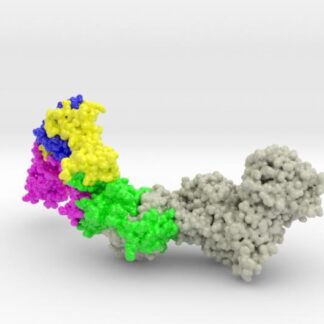
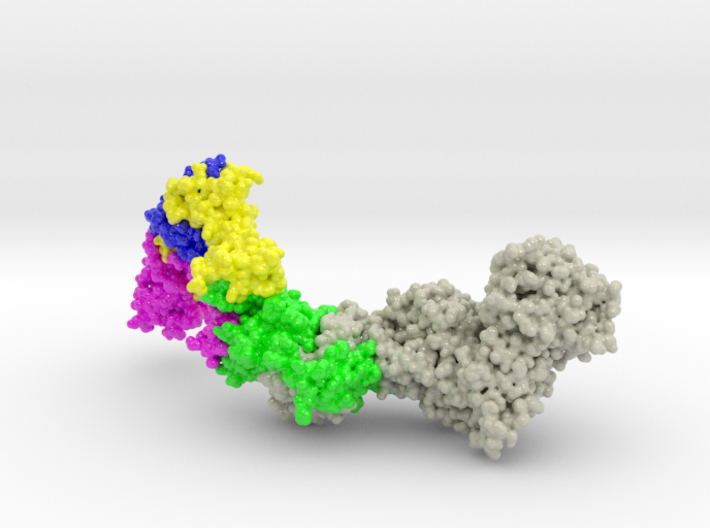
HCMV Pentamer Complex ( gH/gL/UL128/UL130/UL131A ) is the main target of immune response in body fluid, and considered a key target candidate for vaccine therapies. The HCMV gH/gL architecture is similar to that of Epstein-Barr virus (EBV) except for amino-terminal extension in gL. This gL Extension forms a subdomain that acts as a docking site for UL128/UL130/UL131A.
HCMV expresses a collection of proteins that inhibit CD4+ and CD8+ T cells, the normal immune responders to infection. US11 was recently identified as a key component of immune evasion blocking the transfer of maternal Antibodies through the placenta to the baby. Neonatal Fc Receptor (FcRn) controls the transport of Antibodies from mother to baby. US11 prevents Antibody binding to FcRn receptors and dislodges the receptor from the surface of the endoplasmic reticulum. This unique evasion tactic has eluded scientists until now. Look for more updates on HCMV and the therapeutic advances designed to prevent viral birth defects. Learn more about this recent discovery at Nature.com
Biologic Explorer: 3C1B
Http iframes are not shown in https pages in many major browsers. Please read this post for details.
Model Description
Biologic model of the Human Cytomegalovirus (HCMV) Pentamer Complex 3D printed in full-color sandstone and created from PDB ID: 5VOB. Model is colored by protein chain. (Chain A gH75: Grey, Chain B gL: Green, Chain C UL128: Pink, Chain D UL130: Yellow, Chain E UL131: Blue)
3D Print Human Cytomegalovirus