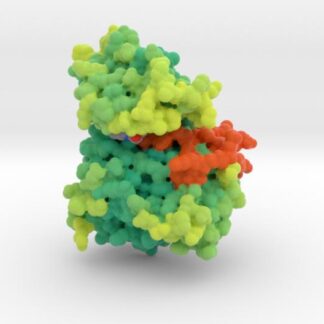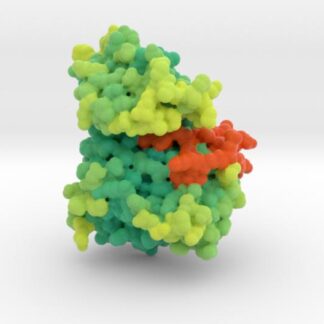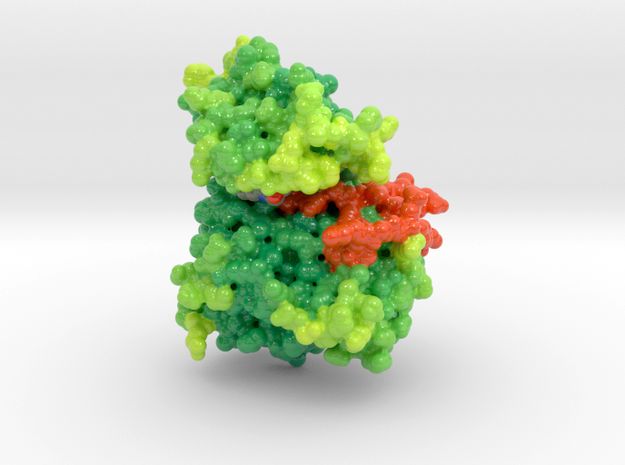
EGFR Mutant Tyrosine Kinase
Mutations in EGFR lead to over-expression of proteins and different types of cancers as cells continually and proliferate. This 3D print of EGFR Tyrosine Kinase visualizes the mutations of key residues that lock the protein’s A-loop (orange) in its active conformation resulting in continued cellular proliferation.
Protein Description
The epidermal growth factor receptor (EGFR) is a transmembrane protein that is a receptor for members of the epidermal growth factor family (EGF family) of extracellular protein ligands. In many cancer types, mutations affecting EGFR expression or activity could result in cancer
Biologic Explorer: 3W2O
Http iframes are not shown in https pages in many major browsers. Please read this post for details.“The epidermal growth factor receptor (EGFR) family plays a critical role in vital cellular processes and in various cancers. Known EGFR inhibitors exhibit distinct antitumor responses against the various EGFR mutants associated with nonsmall-cell lung cancer. The L858R mutation enhances clinical sensitivity to gefitinib and erlotinib as compared with wild type and reduces the relative sensitivity to lapatinib. In contrast, the T790M mutation confers drug resistance to gefitinib and erlotinib. ”
Mutant EGFR
Mutations in EGFR lead to overexpression of proteins and different types of cancers as cells continually and proliferate. Advancements in drug design allow for the continued binding of small molecules despite mutations. These compounds prevent kinase phosphorylation and down-regulate EGFR signals.
Inflammatory Disease
Cancer isn’t the only issue. Psoriasis, eczema, and atherosclerosis are all implicated in over-expression of EGFR. This is not well understood yet, but likely due to over-expression of inflammatory cytokines that initiate EGFR phosphorylation.
Drug Therapies
A common strategy of small molecule inhibitors is to identify receptors and examine their intra-cellular kinase domains. In many cases, this prevents aberrant behaviors in the receptor and mitigate the develop of disease. An easy target for this inhibition process is the kinase domain’s ATP binding pocket. It has a high affinity for its ligand and phosphorylation of its tyrosine residue a requirement for signal propagation.
3D Print EGFR Mutant Tyrosine Kinase