RSV Fusion F Glycoprotein
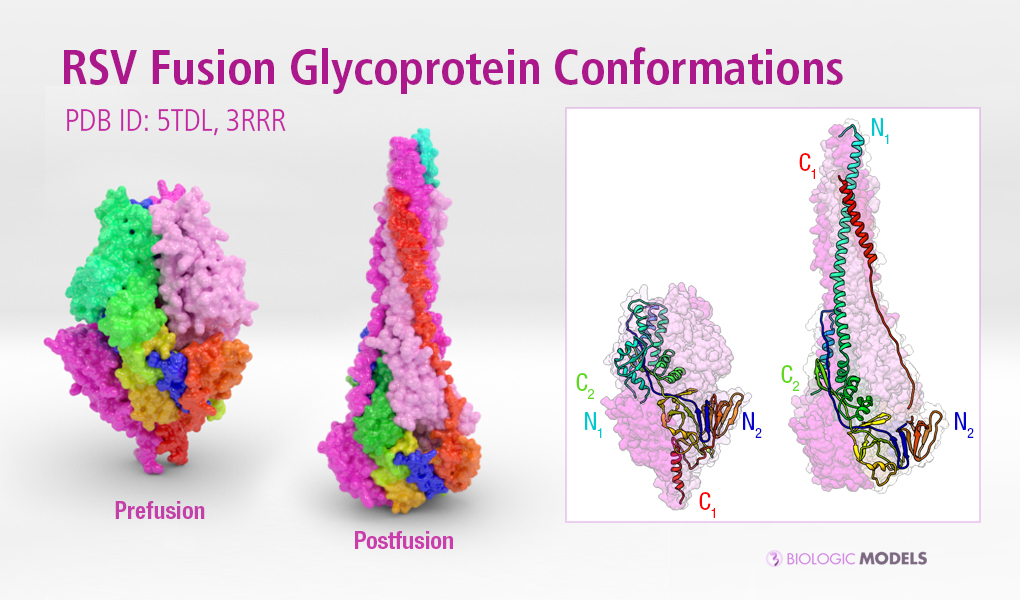
Deadly to newborn babies, the Human Respiratory Syncytial Virus (HSV) is a major cause of lower respiratory tract infection. Dynamic structural changes in the Fusion F Glycoprotein reveal and hide binding sites, making targets of new therapies difficult to identify. Explore RSV Fusion F Glycoprotein-3RRR and learn more about its Postfusion 3D structure.
Protein Description
Respiratory syncytial virus (RSV) is the most common cause of acute lower respiratory infection among children worldwide and the leading cause of infant hospitalization for respiratory disease in developed countries. RSV invades host cells via a type I fusion (F) glycoprotein that undergoes dramatic structural rearrangements during the fusion process. Neutralizing monoclonal antibodies, such as 101F, palivizumab, and motavizumab, target two major antigenic sites on the RSV F glycoprotein.
RSV Fusion Glycoprotein Realtime-3D Biologic Explorer
Use the interactive realtime 3D Biologic Explorer to visualize the internal anatomy and dynamic shape changes of the RSV Fusion Glycoprotein. (Best viewed in a full-screen desktop browser).
3D Print RSV Fusion F Glycoprotein
3D printed protein models of RSV Fusion F Glycoprotein visualize the conformational changes of this viral protein during membrane fusion. Each chain is colored uniquely, with one chain colored blue to red from N to C-Terminal. Using the interface below, configure the scale and materials using the horizontal slider and drop-down menu. Created from PDB ID: 5TDL, 3RRR