Oligosaccharyltransferase Complex
ETH Zurich makes major breakthrough in diabetes research after crystallizing Oligosaccharyltransferase (OST) Complex, the sugar attaching protein.
Whenever cells physically interact or exchange chemical signals, proteins with specific 3-dimensional structure find stable contact with each other. Only until recently, a new and important component of this molecular handshake is the ability of proteins to maintain this connection. Chains of sugar molecules (oligosaccharides) are attached to proteins. Sugar is extremely reactive, finding binding residues throughout the genome. When proteins come into contact, sugar chains attach one protein to another prolonging its the cellular communications signal. If protein-to-protein interactions are a molecular handshake, then oligosaccharides are protein handcuffs binding proteins to each other. The Oligosaccharyltransferase Complex (OST) is the protein that arms newly synthesized proteins from the nucleus with sugar chains.
Back on the Sugar Chain Gang
Anytime our proteins interact with a microbiome, other cells, molecular signals, or frankly do anything, chains of sugar molecules provide important molecular scaffolding necessary to sustain interactions. These sugar appendages dramatically change the conformation of the protein changing its function.
Like a bent sprint losing potential energy, excessive exposure to sugar molecules often damages proteins. Sugar molecules, or monosaccharides, will cause the protein to change shape, preventing it from fully achieving its conformational structure and molecular function. The amount of these sugar molecules attached to cell surfaces is well known to define states of disease and health. (Check out our Hemoglobin A1c Explanation and learn how sugar binds to Hemoglobin).
It’s been known for awhile that sugar chains attach gangs of proteins to each other. Less well understood was how sugar chains first attach to newly synthesized proteins. ETH Zurich asked the question, How do sugar chains get attached? Read More about their work: RESEARCH.
Media Gallery
Protein Description
Oligosaccharyltransferase Complex (OST) is an enzyme located in the cytoplasm of eukaryotic cells on the surface of the endoplasmic reticulum (ER). The ribosomes attached to the ER synthesize new proteins from RNA coming out of the nucleus. Once transcribed, proteins begin folding and require the OST Complex to attach sugar chains in order for the protein to fold correctly.
Protein Structure
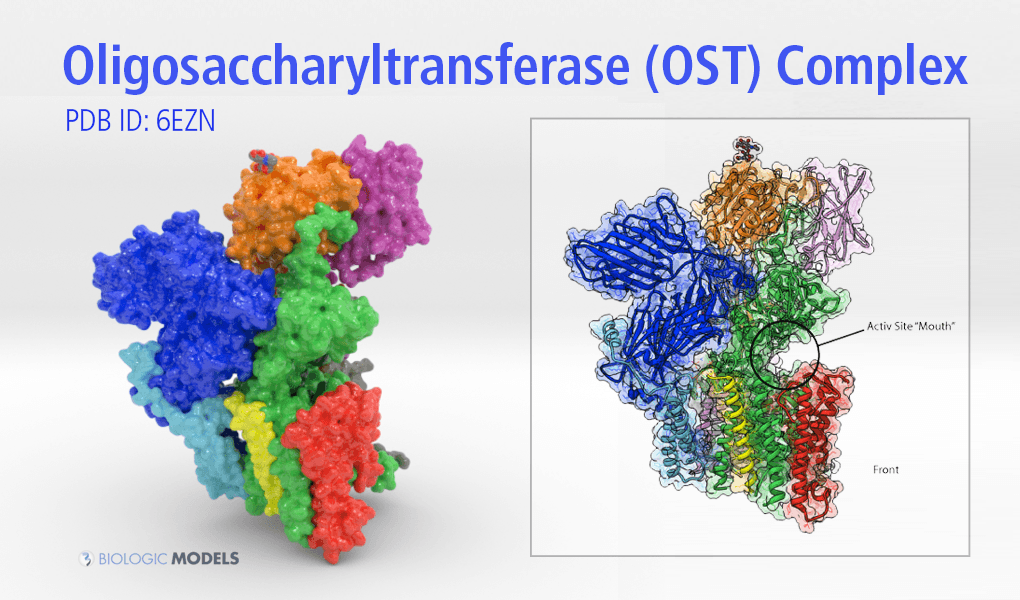
OST is a multichain protein composed of 8 chains(OST1-5, STT3, WBP1, and SWP1). Located just above the transmembrane domains, facing away from the center of the protein in the STT3 chain are its active sites. Captured in this dataset are the actual sugar chains, attached near the active sites providing necessary clues as to how OST glycosylated proteins.
Protein Function
OST is thought to require a multitude of chains due to the complex processes required for both OST assembly and function. These chains must first position themselves near the pores of the endoplasmic reticulum. Then, OST must bind to an oligosaccharyldolichol carrying the sugar chain. Newly folding proteins entering the ER must then be scanned by OST and folding into the proper conformation. After donating the sugar chain provided by the oligosaccharyldolichol, OST finally releases both substrates.
Model Description
This is a 3D printed protein model of Oligosaccharyltransferase Complex. Each chain is colored to match the illustrations from the academic paper published by ETH Zurich, Structure of the yeast oligosaccharyltransferase complex gives insight into eukaryotic N-glycosylation.
Of particular interest to ETH’s research are the active sites (dark green) of OST, found in the green chain F, STT3. The active sites (next to the red chain) are orientated away from the core center of the protein positioned to interact with emerging proteins from the endoplasmic reticulum. Colored by atom type are the monosaccharide glycans.
3D Print Oligosaccharyltransferase Complex