Description
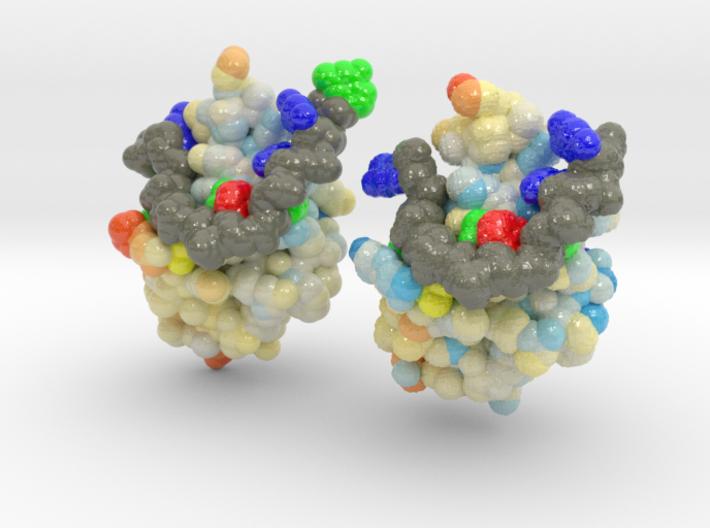
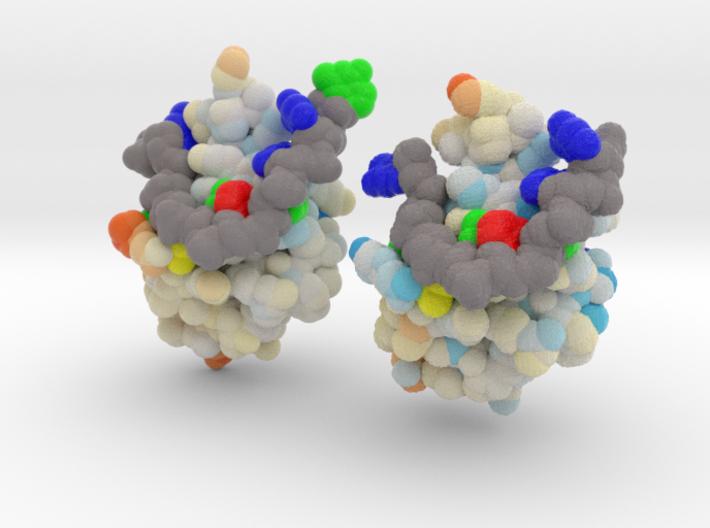
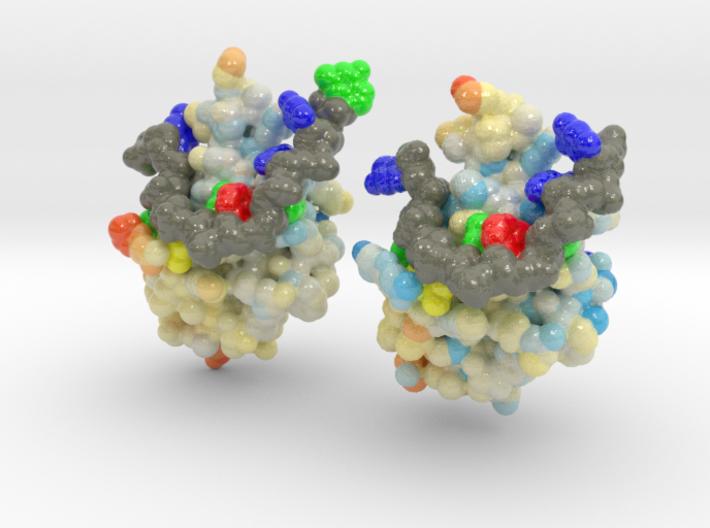
ALS is a fatal motor neuron disorder characterized pathologically by TAR DNA binding protein (TDP-43). The function of TDP-43 is uncertain, and role in ALS remains speculative. Genome scanning reveals mutations associated to neural apoptosis. While TDP-43 mutations have been identified, how these mutations are linked to ALS remains unclear. One of the mutants, D169G, had higher thermal stability than wild-type TDP-43 and that it was cleaved by caspase 3 more efficiently, producing increased levels of the C-terminal 35 kD fragments (TDP-35) in vitro and in neuroblastoma cells. The crystal structure of the TDP-43 RRM1 domain containing the D169G mutation reveal that the D169G mutation induces a local conformational change in a β turn and increases the hydrophobic interactions in the RRM1 core, thus enhancing the thermal stability of the RRM1 domain.
Model Description
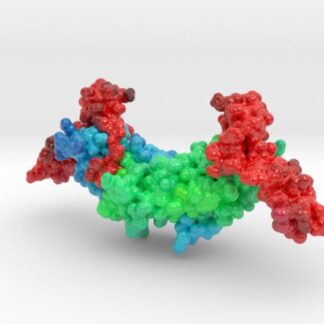
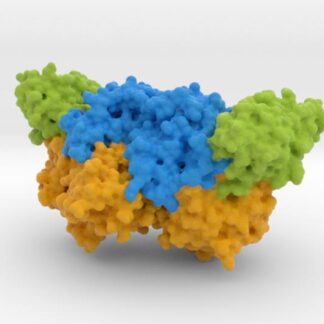
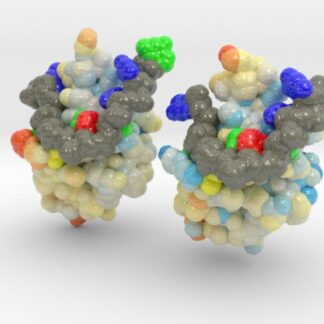
This is a 3D printed model of Human TDP-43 Mutant and Wild Type in Complex with Single-Stranded DNA. TDP-43 mutations are associated with ALS yet it remains unclear how mutations impact function. TDP-43 is colored blue, and the DNA chain backbone yellow. Each nucleotide is colored uniquely (Adenine: red, Cytosine: yellow, Guanine: green, Thiamine: blue).
Created from PDB ID: 4y0f