Description
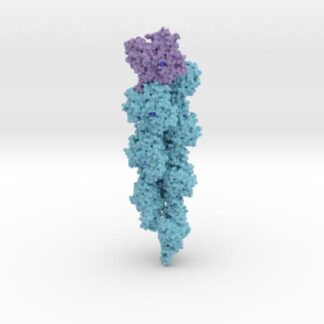
Model Description
Biologic Model of SARS-CoV-2 Spike Glycoprotein Antibody Complex 6VXX-7KS9 is 3D printed and colored coded to match SCIENCE publication denoting functional domains (NTD blue, RBD green, SD1, light tan, SD2 red-orange-yellow, FP cyan, RRAR brown) and the neutralizing human antibody (tinted purple).