Description
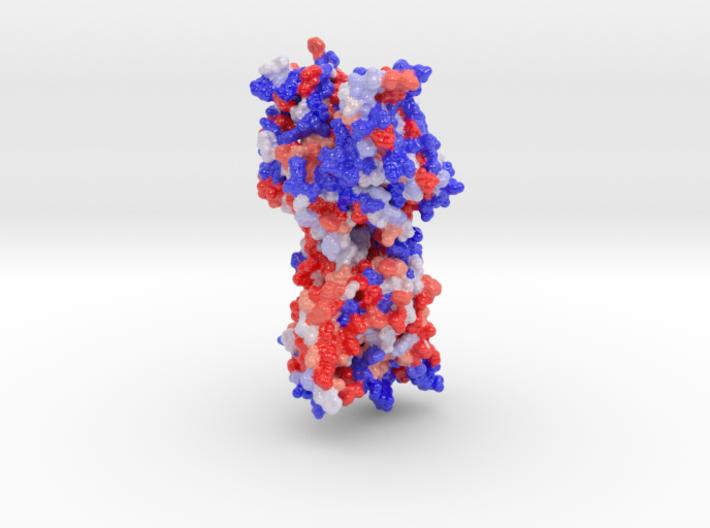
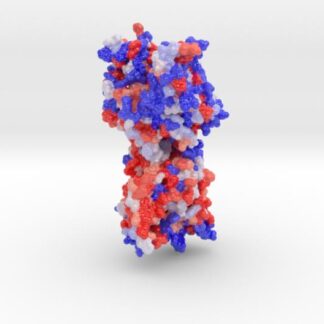
Scientific model of Niemann-Pick C1, 3D printed in full-color sandstone visualizing the protein’s hydrophobicity. 3D printed Biologic Model created from PDB ID: 5i31.
Protein Description
Niemann-Pick C1 protein (NPC1) is a late-endosomal membrane protein involved in trafficking of LDL-derived cholesterol, Niemann-Pick disease type C, and Ebola virus infection.
For international customers outside the US, please visit the model on our 3D printing service’s international website: LINK
Select the desired material finish and size below. Matte finish applies a UV protective, semi-glossy coating. Natural finish does not.