Description
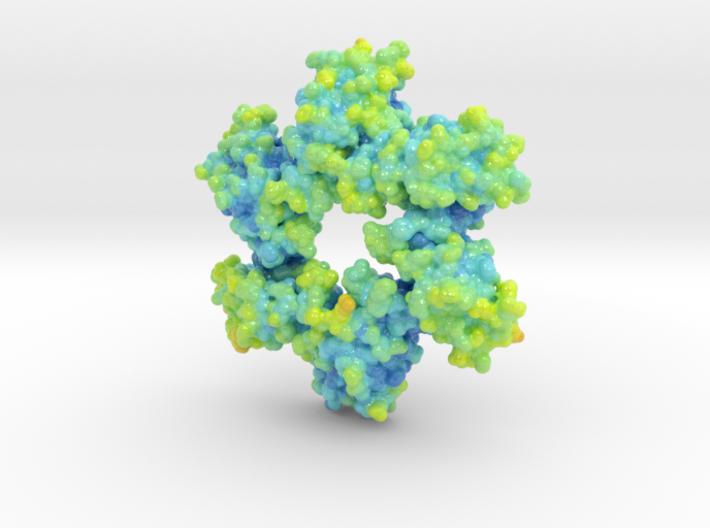
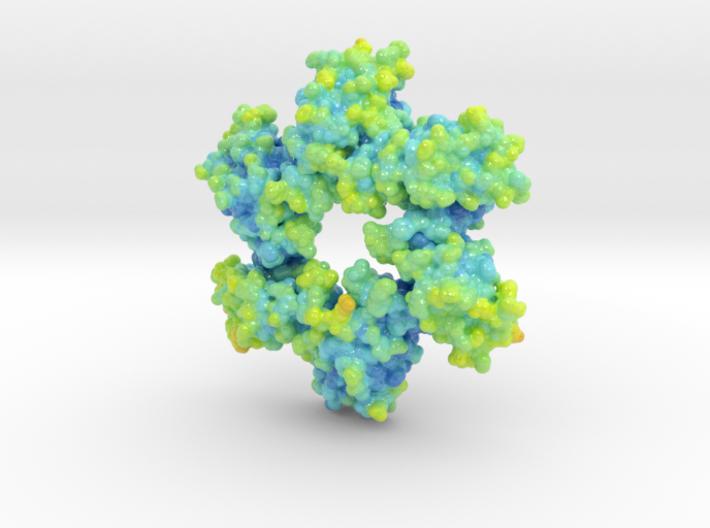
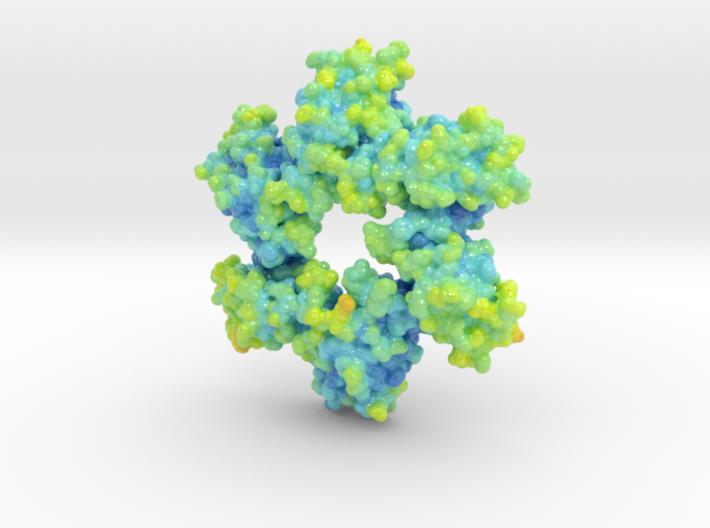
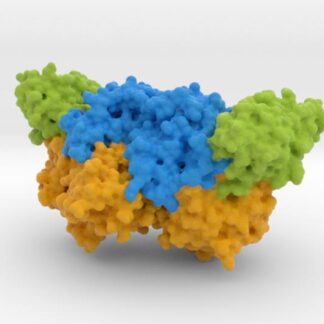
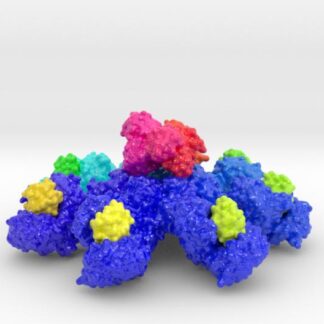
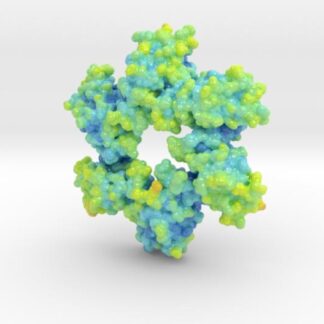
This is a biologic model of Calmodulin-dependent Protein Kinase II 5IG3 3D printed as a volumetric surface to visualize the hexamer’s binding properties. Colored from blue to orange by the protein’s atomic temperature and printed in full-color sandstone.
PubMed Abstract:
Calcium signaling is crucial for several aspects of plasticity at glutamatergic synapses. Synapse activation triggers the exchange of subunits in Ca(2+)/calmodulin-dependent protein kinase II (CaMKII), an oligomeric enzyme that is critical for learning, memory, and cardiac function. The mechanism by which subunit exchange occurs remains elusive. We show that the human CaMKII holoenzyme exists in dodecameric and tetradecameric forms, and that the calmodulin (CaM)-binding element of CaMKII can bind to the hub of the holoenzyme and destabilize it to release dimers. The structures of CaMKII from two distantly diverged organisms suggest that the CaM-binding element of activated CaMKII acts as a wedge by docking at intersubunit interfaces in the hub. This converts the hub into a spiral form that can release or gain CaMKII dimers.
For international customers outside the US, please visit the model on our 3D printing service’s international website: LINK
Select the desired material finish and size below. Matte finish applies a UV protective, semi-glossy coating.
Created from PDB ID: 5IG3